Medical Temperature-Controlled Shipping from China: A Practical Guide for Importers (2026)
Most delays in medical cold chain shipments from China are not caused by the cold chain failing. They are caused by missing documents, wrong packaging, or a forwarder who handles pharma cargo the same way they handle frozen food. By the time you find out, the cargo is sitting in a bonded warehouse at the destination, and the clock is running.
China supplies a significant share of the world's active pharmaceutical ingredients (API), finished dosage products, in-vitro diagnostic (IVD) kits, and temperature-sensitive medical devices. If you source any of these products, this guide covers what you actually need to plan before booking the shipment: the right transport mode, what your documents need to include, what it costs, and where most importers run into problems.
Does Your Medical Product Need Temperature-Controlled Shipping from China?
This sounds obvious, but it is worth confirming before anything else. Not every medical product needs a reefer container or a pharma air freight lane. The storage condition on the product's regulatory registration or product label is your reference point, not what the supplier tells you verbally.
The four temperature ranges that matter for import planning:
2°C to 8°C (refrigerated): Vaccines, finished biologics, insulin, most IVD reagents. Requires continuous refrigeration throughout transit.
15°C to 25°C (controlled room temperature, CRT): Many oral tablets and capsules, some diagnostic kits. Needs temperature monitoring but not active refrigeration in most cases.
-20°C (frozen): Plasma products, certain reagents. Requires a frozen reefer container or insulated packaging with dry ice.
-60°C to -80°C (ultra-low temperature): mRNA products, cell and gene therapy materials. Air freight with specialized active containers is typically the only viable option.
One category that often gets missed: temperature-sensitive medical devices. Certain implantables, biological wound care products, and point-of-care diagnostic kits have storage requirements that are just as strict as pharmaceutical products, but the shipping is sometimes booked as general cargo. Check the device's storage specification before assuming standard freight handling is sufficient.
Air Freight vs. Sea Reefer for Pharmaceutical Cold Chain Shipping
The answer depends on your product's stability window, meaning how long it can safely remain at its storage temperature. Transit time by sea from China to most markets ranges from 20 to 40 days. Air freight runs 2 to 5 days door-to-door.
Air freight is the right choice when:
Your product has a short shelf life or limited stability at storage temperature
The shipment is high-value relative to its weight
You need speed due to clinical, commercial, or regulatory deadlines
The product requires ultra-low temperature (-60°C to -80°C)
Sea reefer is viable when:
Your product's stability allows 30 to 45 days total transit time, including customs clearance at destination
You are importing bulk quantities, such as API or large device batches
Cost per unit matters more than transit speed
If you are new to sea reefer bookings, our guide on how to book a reefer container from China covers the booking process, container specifications, and pre-cooling requirements in detail.
A point importers sometimes overlook: sea reefer containers are not automatically pharma-compliant. The refrigeration unit keeps the temperature within range, but whether the full temperature record meets your destination market's import requirements is a separate question. If your buyer, hospital, or regulatory authority requires documented proof that temperature never exceeded the specification during transit, confirm your forwarder provides that record before booking.
Why Dry Ice Makes Your Medical Shipment a Dangerous Goods Consignment
If your product ships frozen and uses dry ice as a coolant, your shipment is also a dangerous goods shipment. Dry ice is classified as UN 1845, Class 9 under international transport regulations, whether by air or sea.
This means the shipment requires:
A Dangerous Goods Declaration completed by a certified DG shipper
Packaging that meets UN certification requirements
Correct hazard labels on the outer carton
Airlines and ocean carriers check for this at acceptance. A shipment presented without the DG declaration is refused, regardless of the medical urgency. This catches importers off guard because the product itself is not hazardous. The dry ice is.
If your freight forwarder does not handle dangerous goods, they cannot manage a dry ice pharma shipment compliantly. You need a forwarder with both cold chain and DG capability under one roof.
Import Documentation Requirements for Medical Temperature-Controlled Cargo
Missing or misaligned documents are the leading cause of customs delays for medical shipments from China. The standard commercial document set is not enough.
For most medical temperature-controlled imports, you will need:
Commercial Invoice and Packing List with the correct HS code and full product description
Certificate of Analysis (CoA) matching the exact batch number on the packing list
Certificate of Origin required by most markets for tariff classification
Temperature monitoring report showing the full transit temperature record; your destination regulatory authority will typically require this for product release
Dangerous Goods Declaration if dry ice is used as coolant
Import permit or product registration confirmation depending on the destination market and product category
MSDS / SDS for API shipments or any product with a chemical component
A few destination-specific points worth knowing:
Europe: Finished pharmaceutical imports require documentation confirming the product was distributed under GDP-compliant conditions. Your forwarder's handling procedures are part of this.
United States: FDA-regulated products may require prior notice filing before arrival.
Middle East (Saudi Arabia, UAE): Product registration with SFDA or MOHAP must be confirmed before the shipment departs. Unregistered products arriving at these markets face customs hold or destruction.
The single most common documentation error is a batch number mismatch between the CoA and the packing list. It sounds like a minor clerical issue, but it is a standard check at destination customs and GDP-audited warehouses. It stops product release until the discrepancy is resolved.
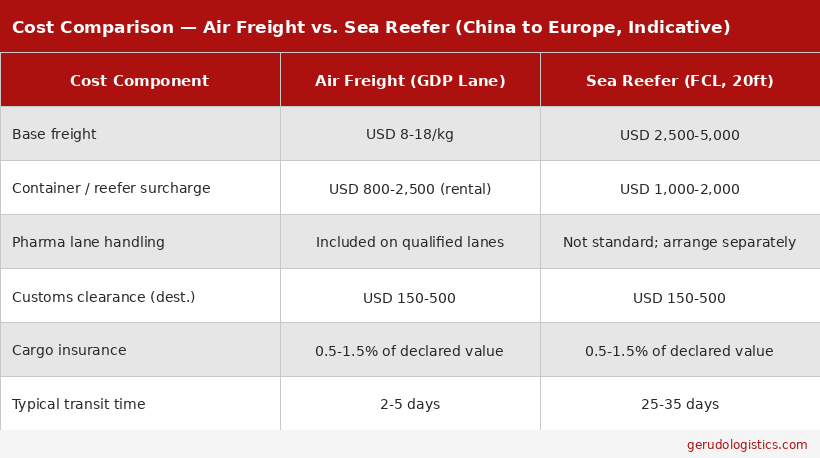
Medical Cold Chain Shipping Costs from China: Air Freight vs. Sea Reefer
Medical cold chain shipping costs more than standard freight, and the gap is larger than many importers expect the first time. The main cost drivers are active container rental for air shipments and reefer surcharges for sea freight, on top of base freight rates.
Air freight reference costs (China to Europe or Middle East):
Base freight on a pharma-qualified air lane: approximately USD 8 to 18 per kg
Refrigerated active container rental (Envirotainer, CSafe, or equivalent): approximately USD 800 to 2,500 per trip, depending on container size
Ultra-low temperature containers cost more than standard refrigerated units
Sea reefer reference costs (China to Europe, 20-foot container):
Base ocean freight: approximately USD 2,500 to 5,000
Reefer surcharge on top of standard dry container rates: approximately USD 1,000 to 2,000
Pre-cooling and data logger costs are additional
These are market reference ranges, not quotes. Actual rates vary by origin port, route, carrier, and timing.
All-risk marine cargo insurance is strongly recommended regardless of mode. Pharmaceutical products often have a replacement cost significantly above their invoice value, particularly biologics or clinical trial materials. Insure for replacement cost, not just the amount on the invoice.
Common Pharmaceutical Cold Chain Shipping Mistakes to Avoid
Based on what we see in practice, the problems that actually affect medical cold chain imports from China fall into a few consistent patterns:
Using a forwarder without DG capability for dry ice shipments. The shipment gets rejected at airline acceptance.
Not confirming product registration at destination before shipping. The cargo arrives and sits in customs because the product is not cleared for import.
Assuming the reefer container is sufficient documentation. The buyer's quality team requires a GDP-compliant temperature record. The container maintained the temperature, but no proper record exists.
Treating the stability window as a buffer. If the product has a 45-day stability window and transit takes 40 days, any customs delay pushes the shipment out of specification.
Batch number errors on documents. CoA and packing list do not match. Product release is held until the issue is resolved in writing.
Most of these are avoidable with proper pre-shipment planning.
How Medical Cargo is Handled in China Before It Reaches You
Most importers focus on what happens after the cargo arrives. The China-side process is where many cold chain failures actually begin.
Step 1: Confirm storage conditions at the supplier's facility
Cold chain integrity starts at the point of storage, not at the port. Ask your supplier to provide a temperature record from their facility covering the period before dispatch. If the product has been sitting outside its required temperature range before pickup, no downstream refrigeration can reverse that.
Step 2: Inland transport from supplier to port or airport
This leg typically uses a refrigerated truck or, for frozen cargo, an insulated vehicle with dry ice replenishment. Confirm with your forwarder whether the vehicle is pre-cooled before loading, whether a temperature monitoring device is on board, and what the contingency is if the vehicle is delayed at a checkpoint.
For air freight, cargo passes through an airport handling facility before loading. The time between truck delivery and aircraft loading can run several hours. Temperature control during this window depends on the ground handler's cold room capacity, which is worth confirming for 2-8°C products.
Step 3: China-side customs clearance
Clearance for pharmaceutical products typically takes one to two days with complete documentation. Common causes of delay include a missing NMPA permit, an HS code mismatch, incomplete DG documentation for dry ice, or discrepancies between the invoice, packing list, and CoA. Any delay here compresses your stability window before the cargo has left China.
Having your forwarder review the full document set before the truck leaves the supplier is the most reliable way to catch these issues early.
Step 4: Port or airport acceptance
For sea reefer, the container is delivered to the terminal one to two days before vessel cut-off and plugged into shore power. Confirm that pre-cooling is completed before cargo is loaded and that a data logger is placed inside before the doors are sealed.
For air freight, DG cargo including dry ice shipments goes through a separate acceptance check. If the DG declaration is missing or packaging does not meet the airline's requirements, the cargo is refused. Rebooking adds at least 24 to 48 hours, during which the product sits unloaded.
Import Customs Clearance for Medical Temperature-Controlled Cargo
Customs clearance for medical cold chain imports takes longer than standard commercial freight in most markets, and the delay directly affects your cold chain. For sea reefer shipments, cargo typically remains in the container at the terminal during clearance. For air freight, the cargo moves to a temperature-controlled pharma handling area if one is available, but this depends on the airport and the ground handler.
What affects clearance time for medical imports:
Product registration status: In regulated markets such as the EU, US, Saudi Arabia, and the UAE, the product must be registered or approved for import before the shipment arrives. Customs will not release unregistered pharmaceutical products regardless of how complete your commercial documents are. Confirm registration status before the shipment departs China.
Import permits: Certain product categories, including narcotics, controlled substances, and some biologics, require a pre-issued import permit. These permits have validity windows, so timing the shipment arrival within that window is part of the planning process.
Duties and taxes: Pharmaceutical products often benefit from reduced tariff rates or exemptions under bilateral trade agreements, but this depends on the correct HS code classification and a valid Certificate of Origin. Incorrect classification delays clearance and can result in higher duty assessments.
Physical inspection: Customs authorities in some markets conduct physical inspections of pharmaceutical imports, particularly for first-time shipments or new suppliers. An inspection typically adds one to three days to clearance time. Factor this into your stability window calculation if the product is close to its transit time limit.
For sea reefer shipments, most destination terminals provide a limited period of free reefer plug-in during clearance. If clearance extends beyond that window, demurrage and additional reefer power charges apply. Confirm the free plug-in period with your forwarder before the vessel departs.
How Gerudo Logistics Handles Medical Temperature-Controlled Shipments
Gerudo specializes in two areas that frequently overlap in medical cargo: cold chain shipping and dangerous goods. When a shipment involves dry ice, the DG compliance and cold chain coordination run through one team rather than two separate vendors. For importers, that means one point of contact and one document set managed consistently from origin to destination.
We operate across China's main shipping ports: Guangzhou, Shanghai, Shenzhen, Ningbo, Qingdao, and Dalian, covering both air and sea freight to major markets including Europe, the Middle East, and North America. On the documentation side, we handle the commercial and logistics document set, DG declarations where applicable, and flag destination-specific import requirements based on HS code and product category.
To discuss a medical cold chain shipment or request a freight quote, contact us today!
Frequently Asked Questions for Pharmaceutical Cold Chain Shipping
Can a standard reefer container maintain pharmaceutical temperature requirements? A reefer container keeps temperature within range, but does not automatically produce a GDP-compliant transit record. Confirm with your forwarder what documentation they provide post-delivery before booking.
What China-side permits are required to import pharmaceuticals from China? Finished pharmaceutical products and certain APIs require NMPA documentation on the China side; your supplier's regulatory team handles this. Confirm it is in order before the shipment is booked, as missing documents at origin cause departure delays.
Does using dry ice mean my shipment is classified as dangerous goods? Yes. Dry ice (UN 1845) is a Class 9 dangerous good, and the shipment requires a Dangerous Goods Declaration, compliant packaging, and correct labelling regardless of whether it travels by air or sea.
What if a temperature excursion occurs during transit? An excursion does not automatically mean rejection: your quality team assesses the magnitude and duration against the product's stability data. If it falls outside the acceptable range, the batch goes on hold pending investigation.
How do I verify that a forwarder is suitable for pharmaceutical cold chain imports? Ask three things: whether they handle DG shipments involving dry ice, whether they provide a full temperature monitoring report post-delivery, and whether they have handled imports to your specific destination market before.
Can temperature-sensitive medical devices be imported by sea from China? Yes, provided the product's stability accommodates 30 to 45 days total transit. Confirm the device's maximum allowable time at storage temperature and assess whether sea transit fits within that window.
How far in advance should I book a medical cold chain shipment? For air freight with active containers, book at least 5 to 7 business days ahead, longer during peak periods. For sea reefer, allow 1 to 2 weeks before vessel cut-off, and start the documentation review earlier than that.
Conclusion
Medical temperature-controlled shipping from China is manageable, but it requires more preparation than standard freight. The product's storage specification drives the mode decision, the mode decision drives the packaging and documentation requirements, and getting any of those steps wrong creates delays that are expensive and sometimes unrecoverable.
The practical checklist before booking: confirm storage requirement and stability window, determine whether dry ice is involved, identify destination market import permit requirements, and verify that your forwarder handles both cold chain and DG under one operation.