How to Ship Cosmetics from China to the US: FDA, MoCRA, and Customs Guide (2026)
The US is one of the most demanding markets for cosmetics imports. Shipping from China to the US involves FDA rules, CBP customs filing, Section 301 tariffs, and specific restrictions for liquid and dangerous goods (DG) products. The rules changed significantly in 2022, and the $800 duty-free threshold for Chinese goods was removed in May 2025.
This guide covers the full import process for the China-to-US route: how to choose a shipping method, what US regulations apply before your goods leave China, and what happens when your shipment arrives at a US port. For a full breakdown of DG classification and documentation from the China side, see our cosmetics shipping guide.
Choosing a Shipping Method for Cosmetics to the US
Three factors determine the right shipping method for the US route: your order volume, whether your products are DG-classified, and how much lead time your inventory cycle allows.
Sea Freight FCL
Full Container Load (FCL) is the most cost-effective option for large orders. Most importers moving full product lines to Amazon FBA or wholesale distribution use FCL.
Transit time: 14-18 days from Shanghai to Los Angeles; 27-33 days to East Coast ports
Add: 3-7 days for customs clearance and inland delivery
Best for: Orders that fill at least a 20ft container
Sea Freight LCL
Less than Container Load (LCL) works when your order does not fill a full container. You share space with other shippers and pay per CBM.
Transit time: 18-25 days to US West Coast ports
Cost: $50-90 per CBM (estimated; DG surcharges apply to liquid and aerosol products)
Note: If your products are DG-classified, confirm that the consolidator accepts dangerous goods before booking
Air Freight
Air freight is the right choice when speed matters more than cost. It suits seasonal launches, high-value products, and urgent stock replenishment.
Transit time: 5-8 days door-to-door
Cost: $6-10 per kg (estimated; rates rise to $9-10/kg during peak e-commerce periods such as Q1 and Q4)
Note: DG liquids like perfumes require more pre-booking checks by air than standard cargo
Express Courier
Express courier (DHL, FedEx, UPS) covers samples, small test orders, and urgent replenishment under 200kg.
Transit time: 3-5 days door-to-door
Cost: $8-15 per kg (estimated)
Note: DG acceptance varies by carrier and product formula. If your supplier quotes express for perfumes or aerosols without asking about DG, verify before booking.
Liquid and DG Cosmetics: What Changes by Mode
Many cosmetics count as dangerous goods (DG) under shipping regulations. This affects which modes are available and what documents must be ready before departure.
Common DG products on the China-to-US route:
Perfumes and alcohol-based fragrances (UN 1266, Class 3 flammable liquid)
Nail polish and nail polish remover (UN 1263 or UN 1090, Class 3)
Aerosol products such as hairspray, dry shampoo, and setting spray (Class 2.1, flammable gas)
Alcohol-based toners and sanitizers with high ethanol content (Class 3)
By sea freight: DG liquids can ship in all container formats. No blanket quantity limits apply to Class 3 consumer goods by sea, though stowage rules do apply. Cargo must be correctly declared, packed in UN-certified packaging, and supported by a valid SDS and DG declaration.
By air freight: Quantity limits are tighter. Under IATA Packing Instruction 307, Class 3 consumer goods such as perfumes are limited to 1 litre per inner packaging on passenger aircraft. Larger volumes require a cargo-aircraft-only designation. Some airlines apply stricter limits on top of IATA minimums, so confirm acceptance before booking.
By express courier: DHL and FedEx accept limited quantities of Class 3 liquids under their DG programs. Some formulas are declined. Always check before booking.
For any DG liquid shipment to the US, documentation must be correct before goods leave China. CBP checks that the physical shipment matches what was declared at origin. Fixing paperwork gaps at a US port is slower and more expensive than catching them before loading.
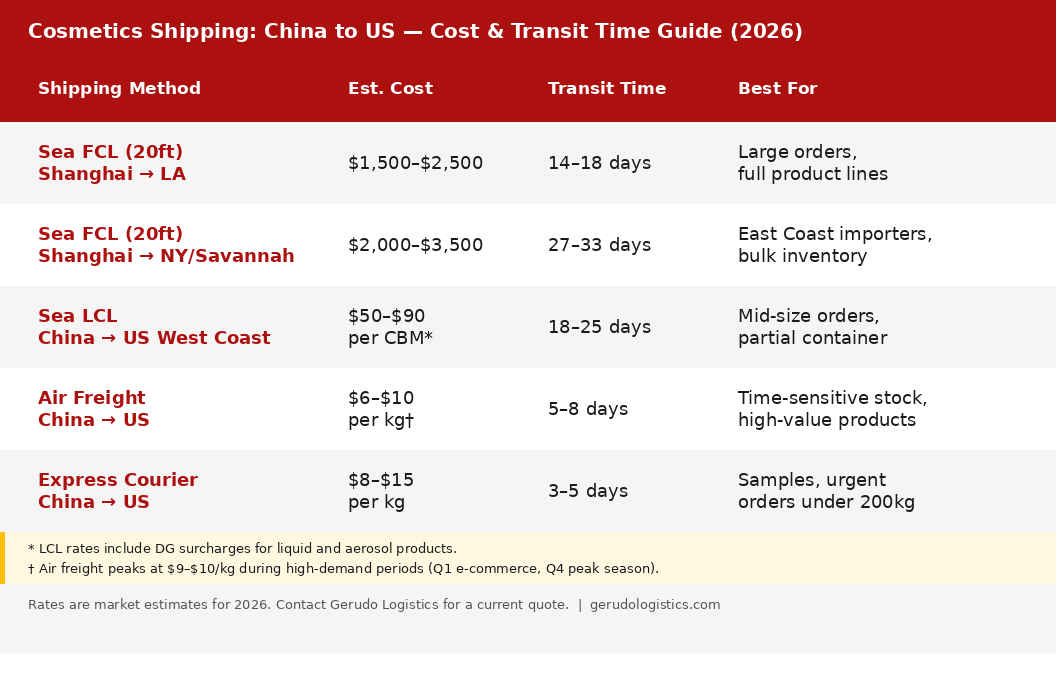
Shipping Cost and Transit Time: China to US
The table below shows estimated freight rates and transit times for the China-to-US route in 2026. Rates vary by port pair, carrier, and market conditions. Contact Gerudo Logistics for a current quote.
Sea freight transit times are port-to-port. Add 3-7 days for customs clearance and inland delivery. DG liquid cosmetics may need extra handling time at origin. Do not book them on the same lead time as standard cargo.
Key US Ports for Cosmetics Imports
Choosing the right port affects transit time, inland freight cost, and how smoothly your DG cargo is processed.
Los Angeles/Long Beach
The largest entry point for Chinese cosmetics. Well-connected to West Coast distribution centers. Most customs brokers handling Chinese consumer goods operate here.
New York/New Jersey
The main East Coast option. Better for importers distributing to northeastern markets or FBA warehouses in that region. Inland costs are lower for customers east of the Mississippi, even though sea transit is longer.
Savannah, Georgia
A growing alternative to New York for East Coast delivery. Shorter port dwell times and good road connections to southeastern logistics hubs.
Seattle/Tacoma
Worth considering for Pacific Northwest distribution or when LA/Long Beach is congested. Transit time from Chinese ports is slightly shorter than Los Angeles for some routes.
For DG cosmetics, confirm with your freight forwarder that your chosen port can handle your cargo type. Most major US ports process DG routinely, but high volumes or specialized formulas may benefit from ports with dedicated hazmat facilities.
US Compliance Requirements for Imported Cosmetics
MoCRA: What Changed in 2022
The Modernization of Cosmetics Regulation Act (MoCRA) was signed into law in December 2022. It is the biggest change to US cosmetics regulation in over 80 years. Its obligations apply directly to importers and foreign manufacturers, including factories based in China.
What MoCRA now requires:
1. Facility registration
Any facility that makes or processes cosmetics for the US market must register with the FDA. This includes factories in China. The deadline for existing facilities was December 2023. If your supplier is not registered, your products may face problems at US customs. Registration is done through the FDA's Cosmetics Direct portal at cosmeticsdirect.fda.gov.
Registration must be renewed every two years. For facilities that completed their first registration before July 2024, the first renewal deadline is July 1, 2026. Confirm that your Chinese manufacturer has completed their 2026 renewal before placing new orders.
2. Product listing
Every product sold in the US must be listed with the FDA. The listing must include the product name, category, and full ingredient information. The responsible person (usually the importer or the US company placing the product on the market) is required to do this.
3. Safety proof
You must be able to show that your products are safe for their intended use. This does not need pre-market FDA approval. But you must have safety data on file. Ask your Chinese manufacturer for safety documentation before you import, not after a problem occurs.
4. Adverse event reporting
If a product you imported causes a serious health problem for a US consumer, you must report it to the FDA within 15 business days.
Cosmetic vs. Drug: Why Claims Matter at Entry
Under US law, the line between a cosmetic and a drug is set by intended use, shown through labeling, packaging, and marketing. Getting this wrong at entry can stop your shipment entirely.
A product is a cosmetic if it: cleanses, beautifies, or changes appearance.
A product may be treated as a drug if it claims to: treat acne, reduce hair loss, lighten skin through active treatment, or offer SPF sun protection.
Why this matters for Chinese suppliers: Supplier marketing language often includes claims that trigger drug classification in the US. A serum labeled "repairs damaged skin cells" or "treats pigmentation" can cause problems at FDA review. Review all label copy and packaging text before placing an order.
US Labeling Requirements
All labels must be in English. Missing or incorrect labels are one of the most common reasons cosmetics from China are detained at US entry.
Required label elements:
Statement of product identity (what the product is)
Net quantity of contents
Name and address of the responsible US party
Full ingredient list using INCI naming, in descending order of predominance
Common labeling failures on Chinese-origin cosmetics:
Ingredient lists using Chinese pharmaceutical names instead of INCI names
Missing net quantity
Incomplete responsible party information
Packaging inserts with claims that suggest drug use
Color additives need separate attention. The FDA has a permitted list, and approval depends on where the product is applied. A color additive approved for body products may not be approved for use near the eye. Color additives that need batch certification must include a batch number on the label.
Prohibited Ingredients, California Prop 65, and De Minimis
Prohibited ingredients under FDA rules include:
Mercury compounds
Bithionol
Chloroform
Vinyl chloride
Halogenated salicylanilides
Certain coal-tar hair dyes
One common mistake: assuming that EU compliance covers FDA requirements. The EU's restricted ingredient list is longer than the FDA's. A product made to EU standards is not automatically FDA-compliant. Always check against FDA rules directly.
California Proposition 65 Prop 65 is a state law, but it covers the largest cosmetics market in the US. Products sold in California that contain listed chemicals above set levels need a clear warning label. Certain fragrances, preservatives, and heavy metals appear on this list. If you distribute to California, confirm Prop 65 status with your manufacturer before goods reach retail.
De minimis no longer applies to China The $800 duty-free threshold for Chinese-origin goods was removed on May 2, 2025. All shipments from China, including small test orders and samples, now go through standard CBP entry and are subject to applicable duties. If you used de minimis for low-cost market testing, you need to update your cost calculations.
US Customs Entry and Duties
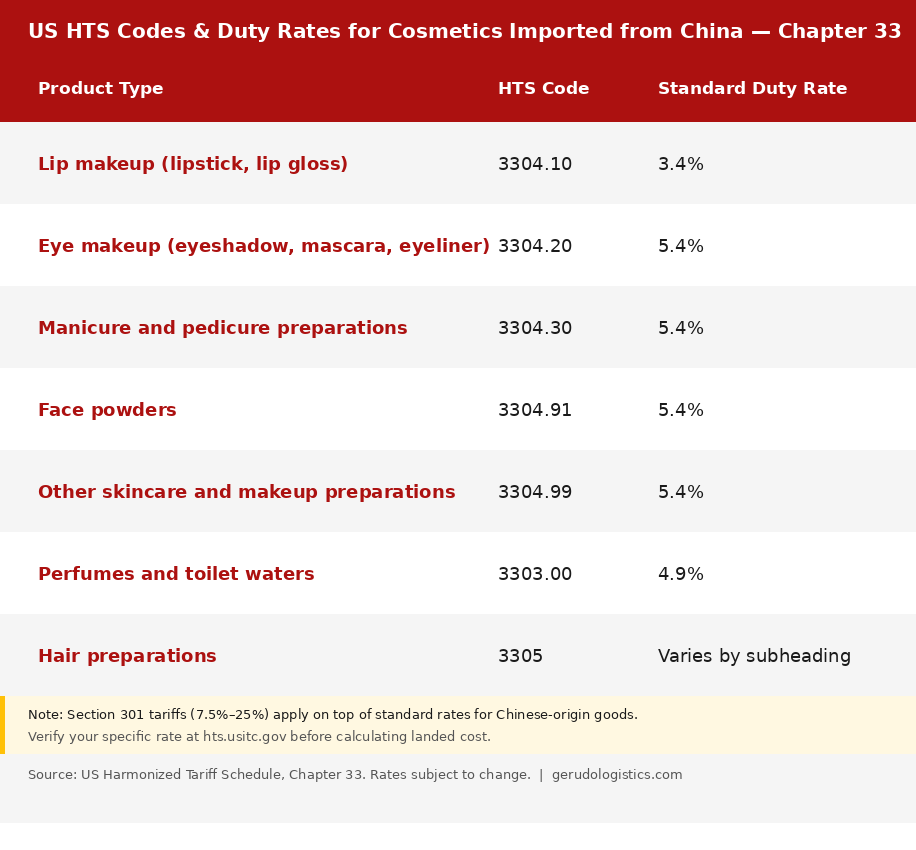
HTS Codes and Duty Rates
Cosmetics from China are classified under Chapter 33 of the Harmonized Tariff Schedule. Standard duty rates are low, but Section 301 tariffs add significantly to the total. Most cosmetics under Chapter 33 face an additional 7.5% to 25% in Section 301 tariffs on top of the base rate.
Some products currently benefit from Section 301 exclusions that reduce or remove the additional tariff. These exclusions are set to expire on November 10, 2026. If your products are on the exclusion list, factor this deadline into your sourcing and pricing plans for late 2026.
Verify the current rate for your specific product at hts.usitc.gov before calculating landed cost. Rates have changed multiple times and may change again.
Other import fees to include in your landed cost:
Merchandise Processing Fee (MPF): 0.3464% of cargo value
Harbor Maintenance Fee (HMF): 0.125% of cargo value (ocean shipments only)
CBP Entry, ISF Filing, and Customs Bond
Import Security Filing (ISF) The ISF must be submitted to CBP at least 24 hours before the vessel leaves the Chinese port. Your customs broker files this, but they need accurate information from you. Wrong product descriptions or HTS codes at this stage create clearance problems that are hard to fix once the ship is at sea.
Customs bond A customs bond is required for any formal entry valued above $2,500. If you import regularly, a continuous bond covering 12 months is more practical than buying a new bond for each shipment.
FDA Review and What Happens with DG Liquids
Most cosmetics shipments pass through FDA review without physical inspection. FDA uses risk-based screening. A shipment may be flagged for:
Prior compliance issues from the same importer
Known problems with a specific manufacturer
Random selection
If a shipment is detained, FDA issues a Notice of Detention and Hearing. You then have a chance to submit compliance evidence. If the issue is a label problem, relabeling under customs supervision may be allowed. If the product itself is non-compliant (wrong ingredients, unapproved drug claims), the only options are re-export or destruction at your cost.
For DG liquid cosmetics, CBP also checks that the physical shipment matches the declared DG classification. If the SDS, UN numbers, or DG declarations are missing or do not match the cargo, the shipment is held. Port storage fees build up quickly. All DG documentation must be in order before goods leave China.
Choosing a China Freight Forwarder for US Cosmetics Imports
Gerudo Logistics specializes in dangerous goods and cold chain shipping from China. For US-bound cosmetics shipments, we handle the parts that most commonly cause problems:
DG documentation review: We check SDS files for Class 2 and Class 3 products before booking and identify gaps that would cause holds at US customs.
Liquid cosmetics compliance: For perfumes, aerosols, nail preparations, and alcohol-based products, we manage DG compliance from packing through port departure.
Customs paperwork: Our team prepares documentation aligned with FDA and CBP requirements, including accurate product descriptions, HTS codes, and ISF coordination.
DDP to US fulfillment centers: We cover freight, customs clearance, duties, and final delivery to Amazon FBA or your warehouse under one confirmed landed cost.
Contact Gerudo Logistics to discuss your US-bound cosmetics shipment and confirm your documentation is complete before departure.
Frequently Asked Questions
Can I ship perfume from China to the US by air?
Yes, within quantity limits. Under IATA Packing Instruction 307, perfumes are limited to 1 litre per inner packaging on passenger aircraft. Larger volumes must travel on cargo-only aircraft. Confirm airline acceptance before booking.
Does my Chinese manufacturer need to be FDA-registered?
Yes. Under MoCRA, any facility making cosmetics for the US market must be registered with the FDA. The deadline for existing facilities was December 2023. Confirm registration status with your supplier before ordering.
What is the cheapest way to ship cosmetics from China to the US?
Sea freight FCL offers the lowest cost per unit for large orders. LCL works for smaller volumes. Air and express are cost-effective when delivery speed justifies the higher rate.
Does the $800 de minimis exemption still apply to Chinese cosmetics?
No. The exemption for Chinese-origin goods was removed on May 2, 2025. All shipments from China now require standard CBP entry and are subject to applicable duties, regardless of value.
What happens if my cosmetics are detained at a US port?
FDA issues a Notice of Detention and Hearing. You can submit compliance evidence. If it is a labeling problem, relabeling under customs supervision may be allowed. Non-compliant products must be re-exported or destroyed.
Do I need California Prop 65 warnings on my products?
If you sell in California and your products contain listed chemicals above threshold levels, warning labels are required. Confirm ingredient status with your manufacturer and apply labels before goods reach retail.
Conclusion
Importing cosmetics from China to the US requires preparation on two fronts: logistics (mode selection, DG handling, port routing, landed cost) and compliance (MoCRA registration, labeling, prohibited ingredients, CBP entry). Problems on either side can hold up your shipment and generate costs that are avoidable with the right groundwork.
The most common issues on this route are DG documentation gaps for liquid products, manufacturers not registered under MoCRA, and labeling that fails FDA review. All three can be identified and fixed before goods leave China.